EDGC pursues the growth of both individuals and organizations,
challenging constantly and cooperating each other.
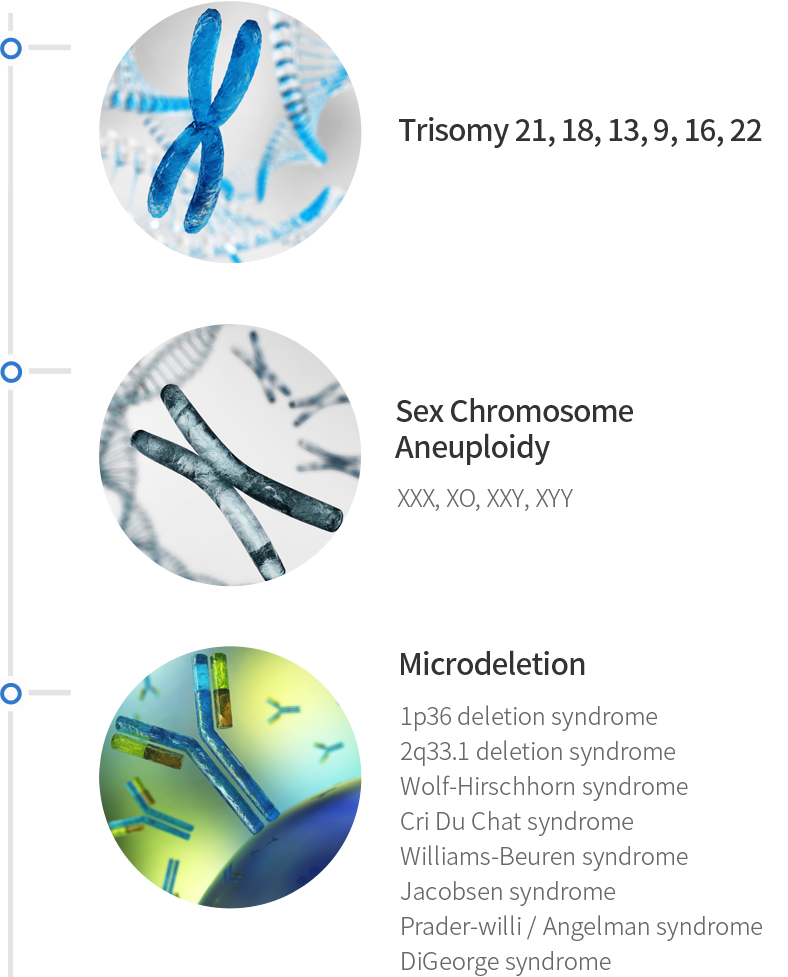
Non-Invasive Prenatal Test (NIPT) detects fetal chromosomal abnormality using fetal DNA in maternal blood with Next Generation Sequencing (NGS).
| Category | Test | Test Method | Test Period | Turnaround Time | Detection Rate |
|---|---|---|---|---|---|
| Non-Invasive Prenatal Test (NIPT) | Non-Invasive Chromosome Examination (NICE®) | Non-Invasive | After 10 Weeks | 7 – 10 Days | Over 99% |
| Blood Screening Test | Triple Test | Non-Invasive | 11 – 13 Weeks | 2 Days | 67% - 71% |
| Blood Screening Test | Quad Test | Non-Invasive | 14 – 22 Weeks | 2 Days | 79% – 81% |
| Integrated Screening Test | Integrated Test | Non-Invasive | 11 – 13 Weeks(1st) | 4 – 5 Weeks | 94% – 96% |
| Integrated Screening Test | Integrated Test | Non-Invasive | 14 – 22 Weeks(2nd) | 4 – 5 Weeks | 94% – 96% |
| Fetal Tissue Screening Test | Chorionic Villus Sampling | Invasive | 10 – 13 Weeks | 4 – 5 Weeks | Over 99% |
| Fetal Tissue Screening Test | Amniocentesis | Invasive | 16 – 21 Weeks | 4 – 5 Weeks | Over 99% |
Positive NIPT cases with confirmatory prenatal karyotype (n=6,238)
| Trisomy | TP(n) | FP(n) | FN(n) | Sensitivity % (95% CI) |
Specificity % (95% CI) |
PPV % (95% CI) |
Accuracy % (95% CI) |
Incidence % (95% CI) |
|---|---|---|---|---|---|---|---|---|
| T21 | 54 | 1 | 0 | 100 (93.40-100) |
99.98 (99.91-100) |
98.18 (88.38-99.74) |
99.98 (99.91-100) |
0.87 (0.66-1.14) |
| T18 | 14 | 0 | 0 | 100 (76.84-100) |
100 (99.94-100) |
100 | 100 (99.94-100) |
0.22 (0.12-0.38) |
| T13 | 2 | 0 | 0 | 100 (15.81-100) |
100 (99.94-100) |
100 | 100 (99.94-100) |
0.03 (0.00-0.12) |
| XO | 6 | 4 | 0 | 100 (54.07-100) |
99.94 (99.84-99.98) |
60 (36.03-79.98) |
99.94 (99.84-99.98) |
0.10 (0.04-0.21) |
| XXX | 1 | 0 | 0 | 100 (2.5-100) |
100 (99.94-100) |
100 | 100 (99.94-100) |
0.02 (0.00-0.09) |
| XXY | 4 | 0 | 0 | 100 (39.76-100) |
100 (99.94-100) |
100 (39.58-100) |
100 (99.94-100) |
0.06 (0.02-0.16) |
| XYY | 1 | 0 | 0 | 100 (2.5-100) |
100 (99.94-100) |
100 | 100 (99.94-100) |
0.02 (0.00-0.09) |
| Total | 82 | 5 | 0 | 100 (95.6-100) |
99.92 (99.81-99.97) |
94.25 (87.23-97.52) |
99.92 (99.81-99.97) |
1.31 (1.05-1.63) |

By acquiring CAP accreditation, EDGC has been recognized with its technology and excellence and made its service reliability higher.

EDGC has acquired the certificate of the Clinical Laboratory Improvement Amendments (CLIA) for Non-Invasive Prenatal Test (NIPT) for the first time in Korea.
| Patent | Country | Date of Registration | Registration Number |
|---|---|---|---|
| Method for Diagnosing Fetal Chromosomal Aneuploidy | South Korea | 2016.09.29 | 10-1663171 |
| Method of Detecting Chromosomal Abnormalities | South Korea | 2018.01.04 | 10-1817180 |
| Novel Method Capable of Differentiating Fetal Sex and Fetal Sex Chromosome Abnormality on Various Platforms | South Korea | 2018.01.05 | 10-1817785 |